Alternative Metabolic Pathways: Gas and Solid-State Fermentation
The landscape of industrial biotechnology is currently undergoing a transformative shift as the limitations of conventional submerged liquid fermentation (SmF) become increasingly apparent in the context of a resource-constrained and carbon-sensitive global economy. While submerged fermentation has long served as the standard for producing antibiotics, organic acids, and various recombinant proteins, the emerging imperatives of circularity and decarbonization have revitalized interest in alternative metabolic platforms, specifically solid-state fermentation (SSF) and gas fermentation (GF).[1, 2, 3] These alternative pathways are not merely substitutes for liquid systems but represent fundamental shifts in how microorganisms interact with their physical and chemical environments to process non-standard feedstocks, ranging from agricultural lignocellulosic waste to industrial C1 off-gases like carbon monoxide and carbon dioxide.[4, 5, 6, 7]
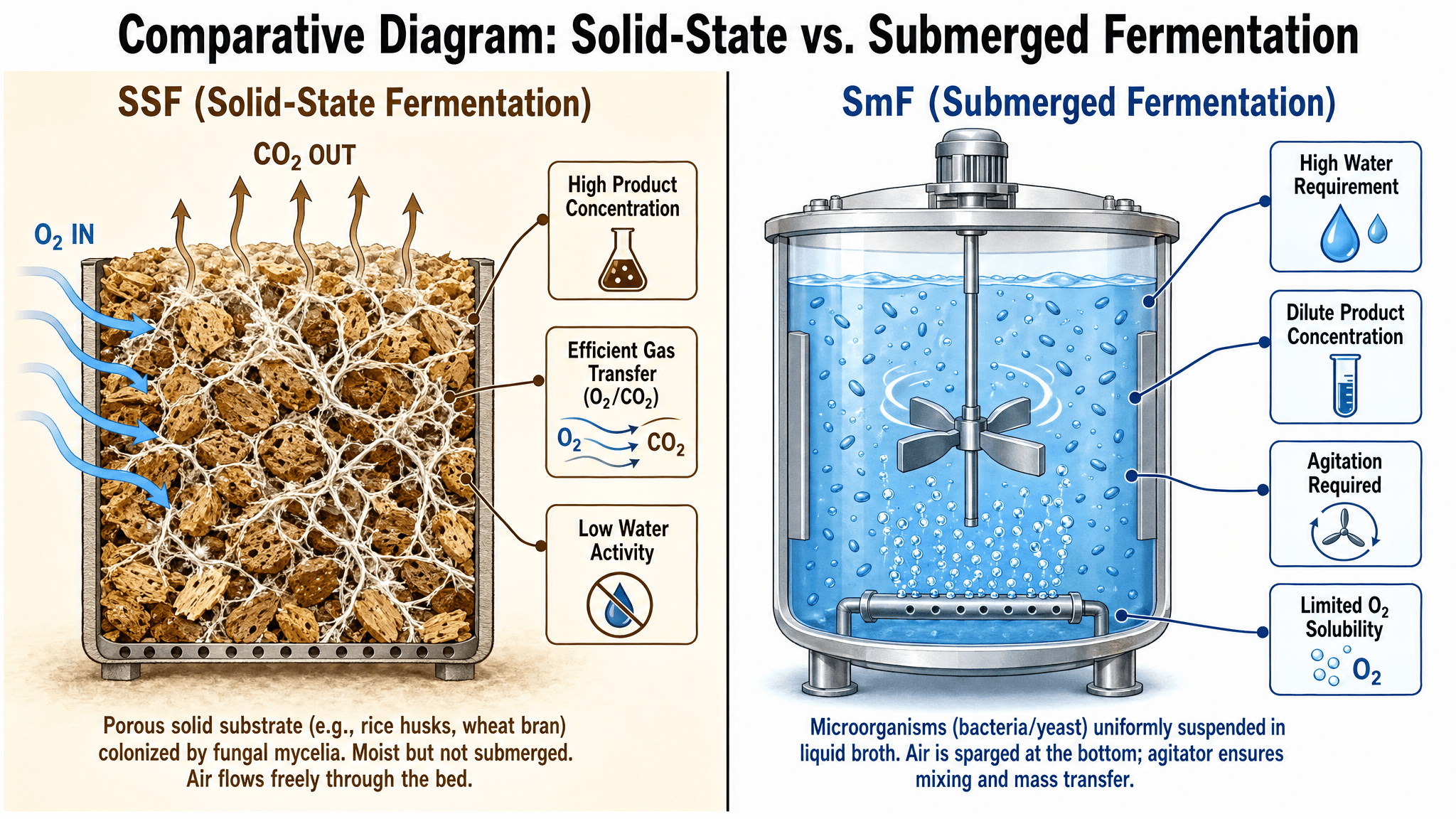
Theoretical Architecture of Solid-State Fermentation
Solid-state fermentation is defined by the cultivation of microorganisms on solid substrates under conditions characterized by the near-total absence of free-flowing water.[1, 2, 4, 8] In this modality, the substrate acts as both a physical scaffold and a nutrient reservoir. The historical context of SSF reaches back to ancient food processing techniques—most notably the production of koji in Asia and mold-ripened cheeses in Europe—but the modern application of this technology has expanded into high-value biomanufacturing.[2, 8] The physical structure of the solid matrix creates a heterogeneous environment that closely mimics the natural habitats of many microorganisms, particularly filamentous fungi, which thrive in the porous spaces of soil and decaying organic matter.[2, 9, 10]
Water Activity and the Bio-Physical Interface
The primary distinguishing factor in SSF is water activity ( ), a thermodynamic parameter that describes the availability of water for microbial metabolism rather than simple moisture content.[4, 10] While submerged systems operate at levels close to 1.0, SSF environments typically maintain values between 0.5 and 0.9.[10] This low-moisture regime exerts a powerful selective pressure; while most bacteria require levels above 0.8 to 0.9 for viable growth, many filamentous fungi are capable of metabolic activity at much lower levels.[10] This differential tolerance naturally mitigates the risk of bacterial contamination, which is a perennial and costly challenge in submerged fermentation, thereby potentially reducing the overhead associated with stringent aseptic processing.[2, 10]
The solid substrate must be carefully prepared to optimize microbial accessibility. Agro-industrial residues such as wheat bran, rice husks, sugarcane bagasse, and fruit peels are the primary feedstocks used in modern SSF.[2, 9, 11, 12] Preparation protocols often involve mechanical grinding to increase surface area, thermal treatments like steaming to break down recalcitrant lignocellulosic barriers, and chemical pre-treatments—such as dilute acid hydrolysis—to liberate fermentable components.[2, 12] These physical modifications are crucial because they determine the porosity of the bed, which in turn governs the efficiency of oxygen diffusion and the dissipation of metabolic heat, two of the most significant engineering constraints in solid-state systems.[4, 10, 13, 14]
Fungal Adaptation and Hyphal Penetration
Filamentous fungi are the preferred biocatalysts for SSF due to their unique hyphal growth morphology.[1, 9, 15] Unlike unicellular yeasts or bacteria that are limited to surface-level interactions in a liquid broth, fungi can penetrate the solid matrix.[1, 11] This “penetrative growth” allows the organism to colonize the interior of substrate particles, reaching nutrients that would be inaccessible in a liquid suspension.[1, 9, 11] The colonization process is facilitated by the secretion of a robust suite of extracellular enzymes that degrade complex polymers into simpler monomers for uptake and assimilation.[8, 9, 16, 17]
|
Feature
|
Submerged Liquid Fermentation (SmF)
|
Solid-State Fermentation (SSF)
|
|---|---|---|
|
Physical Matrix
|
Homogeneous liquid broth
|
Heterogeneous solid particles
|
|
Water Requirement
|
High (free-flowing water)
|
Low (absorbed moisture) [1, 4]
|
|
Microbial Preference
|
Bacteria and Yeasts
|
Filamentous Fungi [1, 13]
|
|
Gaseous Transfer
|
Limited by oxygen solubility
|
High through interstitial voids [4, 14]
|
|
Contamination Risk
|
High (aqueous environment)
|
Low ( selective pressure) [2, 10]
|
|
Environmental Load
|
Significant wastewater volume
|
Minimal effluent generation [2, 18]
|
|
Product Concentration
|
Dilute (high purification cost)
|
Concentrated (reduced downstream) [2, 4]
|
Extracellular Enzyme Dynamics and Metabolic Resilience
A defining advantage of SSF is the ability to bypass traditional metabolic regulatory bottlenecks, most notably catabolic repression.[19] In submerged fermentation, high concentrations of readily metabolizable sugars, such as glucose, often trigger a repression of the genes responsible for producing complex hydrolytic enzymes.[19, 20, 21] This necessitates complex feeding strategies to keep sugar concentrations low while still supporting high biomass growth.[19]
Catabolic Repression and Induction in Solid Beds
Solid-state systems demonstrate a remarkable resilience to catabolic repression. For example, research into Aspergillus niger has shown that enzyme titers for products like pectinases and invertases remain high even when glucose concentrations reach 100 g/L in a solid substrate.[19] In contrast, a concentration of only 30-40 g/L in a submerged system typically results in significant repression.[19] This phenomenon is likely due to the diffusion limitations within the solid bed, which prevent the “flooding” of the fungal cell with glucose, thereby maintaining a balanced metabolic demand even when the total sugar availability is high.[19]
The production of these enzymes is typically induced by the specific chemical signals present in the substrate.[11, 16, 19, 21] Fungi act as sophisticated environmental sensors, modulating their metabolic output based on the complexity of the carbon sources they encounter.[19] Common induction/substrate pairings include:
- Wheat Bran and Amylases: Starch components in cereal residues induce the secretion of -amylase and glucoamylase.[2, 12, 15]
- Coffee Husks and Cellulases: The lignocellulosic nature of coffee waste promotes the expression of endoglucanases and -glucosidases.[22, 23]
- Chitin and Chitinases: The addition of chitin to the solid bed acts as a structural inducer for the production of hydrolytic proteins used in biocontrol.[11]
- Tannic Acid and Tannase: A. niger produces high levels of tannase in response to tannic acid, although concentration limits exist; exceeding 10% tannic acid can lead to a dramatic decrease in enzyme production due to inhibitory effects.[19]
Enzyme Secretion and Kinetic Profiles
The kinetics of enzyme production in SSF often outpace those in SmF. Invertase, for instance, reaches maximum secretion at 24 hours in a solid-state system, whereas it takes 36 hours in a liquid broth.[19] Furthermore, some enzymes that remain associated with the microbial mycelium in submerged cultures are readily excreted into the extracellular space during solid-state growth.[19] This earlier and more robust excretion simplifies the recovery process, as the enzymes are found in a highly concentrated form within the moisture film surrounding the substrate.[2, 4, 11]
|
Enzyme Category
|
Common Industrial Substrates
|
Functional Role in SSF
|
|---|---|---|
|
Cellulases
|
Wheat straw, corn stover
|
Breakdown of cellulose into glucose [8, 9, 11, 16]
|
|
Xylanases
|
Rice bran, sugarcane bagasse
|
Degradation of hemicellulose; biobleaching [8, 9, 11, 17]
|
|
Ligninases
|
Wood shavings, cotton cake
|
Oxidative degradation of lignin via LiP, MnP, or Laccase [8, 17, 24]
|
|
Proteases
|
Soybean meal, cotton cake
|
Hydrolysis of proteins into bioactive peptides [8, 9, 18, 24]
|
|
Amylases
|
Wheat bran, cassava peel
|
Conversion of starch to fermentable sugars [2, 9, 12, 15]
|
|
Lipases
|
Oil cakes, fruit peels
|
Hydrolysis of fats; flavor and aroma production [9, 12, 15]
|
Gas Fermentation: Harnessing C1 Feedstocks
While SSF addresses the valorization of solid organic waste, gas fermentation (GF) offers a solution for the direct bioconversion of gaseous carbon sources.[3, 5, 7] This technology utilizes a group of anaerobic microorganisms known as acetogens to convert carbon monoxide (CO), carbon dioxide ( ), and hydrogen ( ) into fuels and platform chemicals.[5, 7, 25] Gas fermentation is a critical component of modern Carbon Capture, Utilization, and Storage (CCUS) strategies, as it can utilize industrial exhaust from steel mills, biomass gasification syngas, or even captured from the atmosphere.[3, 26, 27, 28]
The Wood-Ljungdahl Pathway: A Metabolic Linear Reducer
The central metabolic framework of gas fermentation is the Wood-Ljungdahl Pathway (WLP), which represents one of the most efficient and ancient routes for carbon fixation.[7, 29, 30] Unlike the circular Calvin-Benson-Bassham cycle used by plants, the WLP is a linear pathway that reduces two molecules of into one molecule of acetyl-CoA.[5, 7, 29]
The pathway is structured into two distinct branches:
- The Methyl Branch: This branch reduces to a methyl group.[5, 29] This process begins with the reduction of to formate by formate dehydrogenase. Formate is then fused to tetrahydrofolate (THF) in an ATP-consuming step catalyzed by formyl-THF synthetase. Subsequent steps involve methenyl-THF cyclohydrolase, methylene-THF dehydrogenase, and methylene-THF reductase to yield methyl-THF.[5] The methyl group is finally transferred to a corrinoid iron-sulfur protein.[5]
- The Carbonyl Branch: In this branch, a second molecule of is reduced to carbon monoxide (CO) by the enzyme carbon monoxide dehydrogenase (CODH).[5, 7, 29] If CO is already present in the feed gas (syngas), it can enter this branch directly.[5]
The enzyme complex acetyl-CoA synthase (ACS) then combines the methyl group from the methyl branch, the carbonyl group (CO), and coenzyme A to produce acetyl-CoA.[5, 7, 29] Acetyl-CoA serves as the primary building block for the synthesis of acetate, ethanol, butanol, 2,3-butanediol, and other high-value bioproducts.[3, 5, 7, 27, 30, 31]
Thermodynamic Limits and Bioenergetic Conservation
Gas fermentation operates at the thermodynamic limit of life.[30, 32, 33] The overall reaction of yields approximately kJ/mol of acetate, which is significantly lower than the energy available from aerobic glucose metabolism.[32, 33] Because the WLP consumes one ATP in the methyl branch and recovers one ATP during acetate formation via substrate-level phosphorylation, there is no net ATP gain from the pathway itself.[5, 33, 34]
To survive and grow, acetogens have evolved specialized respiratory complexes that generate an ion gradient across the cell membrane to power ATP synthesis.[32, 34, 35, 36]
- The Rnf Complex: This membrane-bound ferredoxin:NAD+ oxidoreductase is the primary respiratory enzyme in many acetogens.[32, 35, 36, 37, 38] It catalyzes the exergonic transfer of electrons from reduced ferredoxin ( ) to , using the released energy to pump ions (either in Acetobacterium woodii or in Clostridium ljungdahlii) out of the cell.[32, 35, 38]
- The Ech Complex: Found predominantly in thermophilic acetogens like Thermoanaerobacter kivui , the energy-conserving hydrogenase (Ech) couples the reduction of protons to with the translocation of ions, establishing the necessary electrochemical gradient for ATP synthesis.[32, 34, 39]
- Electron Bifurcation: This recently discovered mechanism allows acetogens to perform thermodynamically “uphill” reactions.[35, 36, 39, 40] In flavin-based electron bifurcation (FBEB), a hydride electron pair is split: one electron is sent to a “downhill” acceptor (like ), while the energy is used to “push” the other electron “uphill” to reduce ferredoxin.[36, 39, 40] This mechanism is essential for regenerating the reduced ferredoxin required for reduction in the WLP.[36, 39, 40]
|
Acetogenic Microorganism
|
Dominant Ion Gradient
|
Respiratory Complex
|
Primary Syngas Products
|
|---|---|---|---|
|
Acetobacterium woodii
|
Sodium ( )
|
Rnf Complex
|
Acetate [32, 34, 35]
|
|
Clostridium ljungdahlii
|
Proton ( )
|
Rnf Complex
|
Ethanol, Acetate [6, 38, 41]
|
|
Clostridium autoethanogenum
|
Proton ( )
|
Rnf Complex
|
Ethanol, 2,3-Butanediol [3, 7, 41]
|
|
Thermoanaerobacter kivui
|
Proton ( )
|
Ech Complex
|
Acetate [32, 34, 39]
|
|
Moorella thermoacetica
|
N/A (Thermophile)
|
Ech/Rnf
|
Acetate, Ethanol [3, 7, 41]
|
|
Clostridium carboxidivorans
|
Proton ( )
|
Rnf Complex
|
Butanol, Hexanol [5, 41]
|
Bioreactor Engineering and Scaling Challenges
The transition from laboratory-scale experiments to industrial-scale production for both SSF and GF is hampered by significant engineering bottlenecks. In both cases, the unique physical nature of the substrates (solids and gases
necessitates reactor designs that move beyond the conventional stirred-tank paradigms of SmF.[1, 3, 4, 10, 13]
Heat and Mass Transfer in Solid Matrices
The most formidable challenge in SSF scale-up is the management of metabolic heat.[4, 10, 14, 42] Because solid substrates have low thermal conductivity compared to water, heat generated by microbial growth can accumulate rapidly within the bed, leading to temperature spikes that inhibit fungal metabolism.[4, 10] Similarly, oxygen gradients can form as the dense substrate bed limits the diffusion of air to the microorganisms located in the interior of the matrix.[10, 14]
To address these issues, several bioreactor configurations have been developed:
- Tray Bioreactors: These are static systems where the substrate is spread in thin layers (1-5 cm) on trays.[10, 13, 14] While they are simple to construct and minimize mechanical stress on the fungi, they suffer from poor heat dissipation in large-scale stacking arrangements.[10, 13, 42]
- Packed-Bed Bioreactors: In these systems, the substrate is packed into a column through which air is forced.[2, 10, 11, 13, 14, 43] Forced aeration significantly improves oxygen supply and convective heat removal, but it can lead to moisture loss and the formation of preferential flow paths (channeling) that result in uneven fermentation.[10, 14, 42, 43]
- Rotating Drum Bioreactors: These involve a horizontal or inclined cylinder that rotates to mix the substrate.[4, 10, 11, 13, 14, 42] The mixing promotes uniform heat and oxygen distribution but can damage the delicate fungal hyphae if the rotation speed is too high, leading to reduced enzyme yields.[10, 14, 42]
- Fluidized-Bed Bioreactors: Substrate particles are suspended by a high-velocity upward flow of air.[10, 13, 14] This provides excellent heat and mass transfer but requires precise control to maintain particle suspension without excessive attrition.[10, 13, 14]
Overcoming Gas-Liquid Mass Transfer Limits
In gas fermentation, the primary technical barrier is the low solubility of and in water.[3, 5] The volumetric mass transfer coefficient ( ) must be high enough to ensure that the rate of gas dissolution matches the microbial uptake rate.[3] This often requires high-energy agitation or specialized gas distribution systems, such as micro-bubble spargers or the use of nanoparticles to enhance gas transport.[3, 25] Furthermore, industrial off-gases often contain impurities like sulfur compounds or hydrogen cyanide that can poison biocatalysts, although acetogens are generally more tolerant to these contaminants than traditional chemical catalysts like those used in the Fischer-Tropsch process.[3, 5, 6]
|
Bioreactor Design Type
|
Primary Advantage
|
Scaling Constraint
|
|---|---|---|
|
Tray (SSF)
|
Minimal shear stress; simple
|
Significant heat gradients [10, 13, 42]
|
|
Packed-Bed (SSF)
|
Efficient forced aeration
|
Bed compaction and channeling [14, 42, 43]
|
|
Rotating Drum (SSF)
|
High uniformity of mixing
|
Mechanical damage to mycelia [10, 14, 42]
|
|
Stirred Tank (GF)
|
Established technology
|
High energy for mass transfer [3, 25]
|
|
Bubble Column (GF)
|
Lower OPEX than stirred tank
|
Lower mass transfer efficiency [3]
|
Techno-Economic Analysis and Environmental Impact
The viability of alternative fermentation pathways is intrinsically linked to their environmental performance and economic competitiveness relative to fossil fuels and conventional bioprocessing.[3, 28, 44, 45]
Comparative Life Cycle Assessment (LCA)
Life Cycle Assessment studies have provided compelling evidence for the environmental benefits of SSF over SmF.[22, 23] For the production of cellulases from coffee husk waste, SSF was found to have a 12.8% lower environmental impact across most categories compared to a submerged process using pure cellulose as a substrate.[23] Specifically, SSF recorded lower global warming potential (425,722 kg eq vs. 492,715 kg eq for SmF) and terrestrial ecotoxicity.[23]
However, the downstream processing (DSP) phase remains a major contributor to the carbon footprint in both technologies.[22, 23] Sensitivity analyses indicate that lyophilization (freeze-drying) is the single most energy-intensive step; removing this step could reduce the environmental impact of SSF by up to 93%.[23] This highlights a critical need for innovations in in-situ product recovery and more energy-efficient drying technologies.[3, 22, 23]
Economic Metrics of Single-Cell Protein and Biofuels
In the domain of gas fermentation, Techno-Economic Analysis (TEA) for single-cell protein (SCP) production has established a minimum selling price (MSP) of approximately $2,070 per metric ton.[28] This calculation assumes the use of wind-powered water electrolysis for generation and captured from corn ethanol production.[28] While this price is currently higher than traditional soybean meal, the environmental metrics are superior: SCP production uses only 0.4% of the land required for soybeans and has a carbon intensity as low as 0.73 kg -equiv/kg protein.[28]
For biofuels, gas fermentation can reduce greenhouse gas emissions by at least 60% compared to fossil gasoline, with biomass-derived syngas pathways reaching up to 90% reduction.[45] The primary economic challenge remains the high CAPEX for syngas generation and the relatively low concentration of products like ethanol in the fermentation broth, which increases the energy requirements for distillation.[3]
|
Metric
|
Single-Cell Protein (GF)
|
Fishmeal (Traditional)
|
Soybean Meal (Traditional)
|
|---|---|---|---|
|
Carbon Intensity
|
0.73 kg eq/kg
|
2.72 kg eq/kg
|
0.85 kg eq/kg [28]
|
|
Land Use
|
0.4% relative to soy
|
Marine-based
|
100% (Reference) [28]
|
|
Ecosystem Impact
|
<0.1% marine disturbance
|
High (Overfishing risk)
|
High (Deforestation/Land use) [28]
|
|
Min. Selling Price
|
$2,070 / metric ton
|
Variable (Market)
|
Variable (Market) [28]
|
Synthetic Biology and the 2024-2026 Horizon
As we move toward 2026, the convergence of synthetic biology and metabolic engineering is providing the tools necessary to overcome the inherent limitations of these alternative pathways.[3, 46, 47, 48]
Engineering the Next Generation of Biocatalysts
Advances in CRISPR-Cas systems and genome-scale metabolic modeling are enabling the design of “chassis” organisms that are optimized for either solid-state or gas environments.[3, 47, 48] In gas fermentation, research is focusing on enhancing the oxygen tolerance of acetogens.[3] For instance, recent studies in 2026 identified the A559 amino acid residue in Carboxydothermus hydrogenoformans as a target for site-directed mutagenesis to create oxygen-tolerant CODH enzymes.[3] This would allow for less stringent anaerobic conditions, potentially lowering the CAPEX of reactor infrastructure.[3]
In solid-state fermentation, synthetic biology is being used to optimize the “secretome” of fungi like Trichoderma reesei and Aspergillus niger.[9, 11, 49] This includes the creation of strains with high-level constitutive expression of enzymes, effectively removing the need for expensive structural inducers.[11, 19, 21] Furthermore, metabolic engineering for methanol utilization is a growing hotspot, with strategies focusing on modifying native pathways and designing artificial assimilation routes to expand the feedstock range of methylotrophic microbial factories.[50]
Commercial Landscape and Funding Realities
The commercial landscape for fermentation-enabled products, particularly alternative proteins, experienced a significant test of assumptions in 2024 and 2025.[49, 51] While the total funding for alt-protein fermentation startups fell from $651 million in 2024 to $357 million in 2025, several key players reached commercial milestones.[49, 51] The launch of whole-cut mycoprotein steaks and eggs produced via precision fermentation in supermarkets like Walmart signals a shift from “promise to proof”.[49, 51]
However, the collapse of some high-profile players has emphasized the risks associated with scale-up pathways.[51] Governments have responded with increased public investment, such as the $51 million allocated by the United States for precision fermentation capacity and the €50 million earmarked by the European Union for biotechnology-driven low-emission food production.[49]
Synthesis and Industrial Implications
The integration of gas and solid-state fermentation into the global industrial fabric represents a fundamental shift toward a bio-based, circular economy. Solid-state fermentation offers an unparalleled opportunity to valorize bulky, low-value agricultural residues into high-value enzymes and proteins with minimal water use and wastewater generation.[2, 8, 18] Its metabolic resilience and resistance to catabolic repression make it an ideal platform for decentralized, waste-integrated biomanufacturing.[3, 19]
Concurrently, gas fermentation provides a versatile solution for the carbon challenge, transforming industrial off-gases and into a diverse array of liquid fuels and functional ingredients.[3, 5] While technical hurdles in mass transfer and economic barriers related to energy costs persist, the environmental mandate and the rapid evolution of synthetic biology tools are driving these technologies toward commercial maturity.[3, 47]
The success of these alternative metabolic pathways will ultimately depend on three pillars: first, the continued engineering of more robust microbial strains that can thrive at the thermodynamic edge; second, the development of novel bioreactor designs that solve the physical constraints of solid and gas substrates; and third, the establishment of policy and carbon-credit frameworks that accurately monetize the superior environmental value of these next-generation bioproducts.[3, 28, 47] As we move toward 2030, the ability to “hijack” native cellular machinery and adapt it to non-standard environments will be a defining capability of the global bioeconomy.[48, 52]
——————————————————————————–
- What are the differences between solid surface fermentation and submerged fermentation? – Quora
- Solid State Fermentation – Part 1: Processes and Application – Niras
- Technological Innovation in Syngas Fermentation and Prospects for …
- Solid-State Fermentation: An Overview – SciSpace
- Bacterial synthesis gas (syngas) fermentation – Taylor & Francis
- Clostridium ljungdahlii represents a microbial production platform based on syngas – PNAS
- Engineered acetogenic bacteria as microbial cell factory for diversified biochemicals
- Solid-state fermentation as a strategy for improvement of bioactive properties of the plant-based food resources – PMC
- Solid-state fermentation—a sustainable future technology in aquafeeds? – Frontiers
- Design Optimization of a Tray Bioreactor for Solid-State Fermentation: Study of Process Parameters through Protein Modification of By-Products – MDPI
- Design of Solid-State Fermentation Systems for Polymer Hydrolytic …
- Optimization of Solid-State Fermentation for High-Yield Enzyme Production Using Agro-Waste Substrates – Walsh Medical Media
- Solid state fermentation – Tech4BioWaste
- Solid State Fermentation – Part 2: Technology Design and Process Utilisation – Niras
- It Is the Mix That Matters: Substrate-Specific Enzyme Production From Filamentous Fungi and Bacteria Through Solid-State Fermentation – PubMed
- Fungal extracellular enzyme activity – Wikipedia
- Extracellular Enzyme Activities and Carbon/Nitrogen Utilization in Mycorrhizal Fungi Isolated From Epiphytic and Terrestrial Orchids – Frontiers
- Optimization of solid-state fermentation conditions for high β-galactosidase-producing lactic acid bacteria and its application in low-lactose dairy products – Frontiers
- Production of enzymes by solid substrate fermentation – Horizon IRD
- Metabolic regulation of fermentation processes | Request PDF – ResearchGate
- Metabolic regulation and overproduction of primary metabolites – PMC – NIH
- (PDF) Solid-State Fermentation (SSF) versus Submerged Fermentation (SmF) for the Recovery of Cellulases from Coffee Husks: A Life Cycle Assessment (LCA) Based Comparison – ResearchGate
- Solid-State Fermentation (SSF) versus Submerged Fermentation …
- Bioactives and Extracellular Enzymes Obtained from Fermented Macrofungi Cultivated in Cotton and Jatropha Seed Cakes – PMC
- Syngas fermentation – Wikipedia
- Untitled
- Wood–Ljungdahl pathway: the process for fermenting CO and CO2 to produce ethanol and acetic acid. ⁹ – ResearchGate
- Techno-Economic Analysis of Gas Fermentation for the Production …
- Wood–Ljungdahl pathway – Wikipedia
- Model acetogens as chassis for CO2-driven bioproduction – PubMed
- Two-stage process and strain engineering for continuous bioconversion of CO2 to butanol | Request PDF – ResearchGate
- 3 Structure – Mikrobiologie Frankfurt
- Overcoming Energetic Barriers in Acetogenic C1 Conversion – PMC
- Energy Conservation in the Acetogenic Bacterium Clostridium aceticum – MDPI
- Molecular principles of redox-coupled sodium pumping of the …
- Flavin-Based Electron Bifurcation, Ferredoxin, Flavodoxin, and Anaerobic Respiration With Protons (Ech) or NAD+ (Rnf) as Electron Acceptors: A Historical Review – Frontiers
- The Rnf Complex Is an Energy-Coupled Transhydrogenase Essential To Reversibly Link Cellular NADH and Ferredoxin Pools in the Acetogen Acetobacterium woodii – PMC
- The Rnf Complex of Clostridium ljungdahlii Is a Proton-Translocating Ferredoxin:NAD+ Oxidoreductase Essential for Autotrophic Growth – PMC
- Molecular Basis of the Electron Bifurcation Mechanism in the [FeFe]-Hydrogenase Complex HydABC – PMC
- Electron Bifurcation: Thermodynamics and Kinetics of Two-Electron Brokering in Biological Redox Chemistry | Accounts of Chemical Research – ACS Publications
- Genetic and metabolic engineering challenges of C1-gas fermenting acetogenic chassis organisms – PMC
- Seven closed solid-state fermentation systems – knikbio.com
- A Critical Evaluation of Recent Studies on Packed-Bed Bioreactors for Solid-State Fermentation – MDPI
- The role of techno-economic and life cycle assessment in guiding precision fermentation towards sustainable food production – ResearchGate
- Life Cycle Assessments of Ethanol Production via Gas Fermentation: Anticipated Greenhouse Gas Emissions for Cellulosic and Waste Gas Feedstocks | Industrial & Engineering Chemistry Research – ACS Publications
- Metabolic Engineering and Synthetic Biology: Synergies, Future, and Challenges
- Synthetic biology and metabolic engineering paving the way for sustainable next-gen biofuels: a comprehensive review – Energy Advances (RSC Publishing) DOI:10.1039/D5YA00118H
- Key Technologies of Synthetic Biology in Industrial Microbiology – PMC – NIH
- 2024 State of the Industry: Fermentation for meat, seafood, eggs …
- Advances in synthetic biology for engineering methylotrophic microbial cell factories
- Funding dip for alt protein fermentation signals shift from promise to proof – AgFunderNews
- Metabolic Engineering and Synthetic Biology for a Sustainable Future